Microbial Ecology. Biogeochemistry.
Stable Isotope Ecology.
Plant-Microbe Interactions.
Ecosystem Response to Change.
Microbiology in Extreme Environments.
|
CURRENT: Microbiology in the Critical Zone: "hot spots and hot moments", and ecosystem services in the Catalina-Jemez Critical Zone Observatory
The JRB-SCM Critical Zone Observatory is an interdisciplinary observatory that will improve our fundamental understanding of the function, structure and co-evolution of vegetation, soils, and landforms that comprise the Critical Zone (CZ). This CZO lead by Jon Chorover has developed a conceptual framework of (a) “hot spots and hot moments”, where landscape change occurs non-uniformly over space and time, for example occurring rapidly after major fire events (which our CZO just experienced in summer 2013), and (b) an energy/carbon flow equation ("EEMT", effective energy and mass transfer, developed by CZO PI Craig Rasmussen) that quantitatively describes critical zone landscape evolution. With the recent summer 2013 renewal the SWES-MEL lab and the Gallery Lab have begun to bridge microbiology and CZO biogeochemistry in the context of water and soil processes, including decomposition, weathering, carbon stabilization, and carbon flow, and water purification. Our approach spans both physiological (Gallery Lab) and meta-omic (Rich lab) approaches. |
|
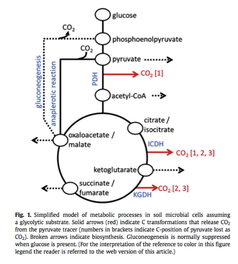
Undergraduate Research: Testing models of central carbon metabolism for soil microbial communities.
I began working for Dr. Paul Dijkstra in the Ecosystems Climate Change and Solutions group during the summer of 2012 at Northern Arizona University. My project investigated soil carbon (C) cycling using mathematical models to predict microbial biochemical processes that contribute to soil C storage and release. Soils hold the largest C pool in terrestrial ecosystems and poor understanding of physiological processes in situ hinders predictions of feedbacks with changing environmental conditions and climate. My experiment tested the accuracy of these metabolic models used to calculate biochemical processes in intact soil microbial communities using stable isotope probing techniques. I presented the results of my experiment at the American Geophysical Union (AGU) conference in December 2012. |
samples used for model testing were taken from open areas along an elevation gradient in northern Arizona from grassland, Juniper-Pinyon and Ponderosa plant dominated sites.
|
|
Undergraduate Research: Testing temperature limits of microbial metabolism in hot spring microbial communities. While I was working on the experiment testing the metabolic flux model during the summer of 2012, I had the opportunity to aid in a weeklong fieldwork trip with Dr. Paul Dijkstra to investigate microbial biogeochemistry at the Great Boiling Hot Springs in Gerlach, Nevada. This work was part of an international collaborative effort that connected professors, field technicians, graduate and undergraduate researchers from Montana State University, University of Nevada, Las Vegas, and Tongjii University. I assisted in field incubation experiments and analyzing samples taken in the field on the Picarro cavity-ring down spectrometer. Our goal was to characterize the biogeochemistry and microbial community dynamics across a temperature gradient in order to test temperature limits of C and N cycling. Paul Dijkstra presented results in a poster at AGU 20129. This experience taught me the value of cross-disciplinary collaboration in addressing microbial dynamics in extreme environments. |
|
Undergraduate Research: Exotic Tamarisk soil legacy effects on exotic and native plant seedling establishment.
I began my research career working in the laboratory group led by Dr. Catherine Gehring at Northern Arizona University who specializes in mycorrhizal fungal relationships with plants. This first experience fueled my passion for creative and critical thinking and I began working on drafting my own independent research project to test whether an exotic species, Russian Olive, would outcompete native Fremont Cottonwoods in soils impacted by exotic Tamarisk. This project originated from previous findings that Tamarisk increased the amount of salt in the soil and disrupted beneficial mycorrhizal fungal mutualisms1. After writing and submitting the grant proposal, I received the Hooper Undergraduate Research Award for $3,500 to finance the project over the course of a year beginning 2012. I conducted a greenhouse study planting Fremont Cottonwood and Russian Olive seedlings in live and sterilized soil collected from a Tamarisk stand. I found that Tamarisk soil increased potentially pathogenic fungal colonization in both species, which had a greater negative effect on the growth of Russian Olive seedlings relative to Fremont Cottonwoods. I presented findings at the May 2013 Hooper Undergraduate Research Award Symposium and the NAU Undergraduate Research Symposium2. |